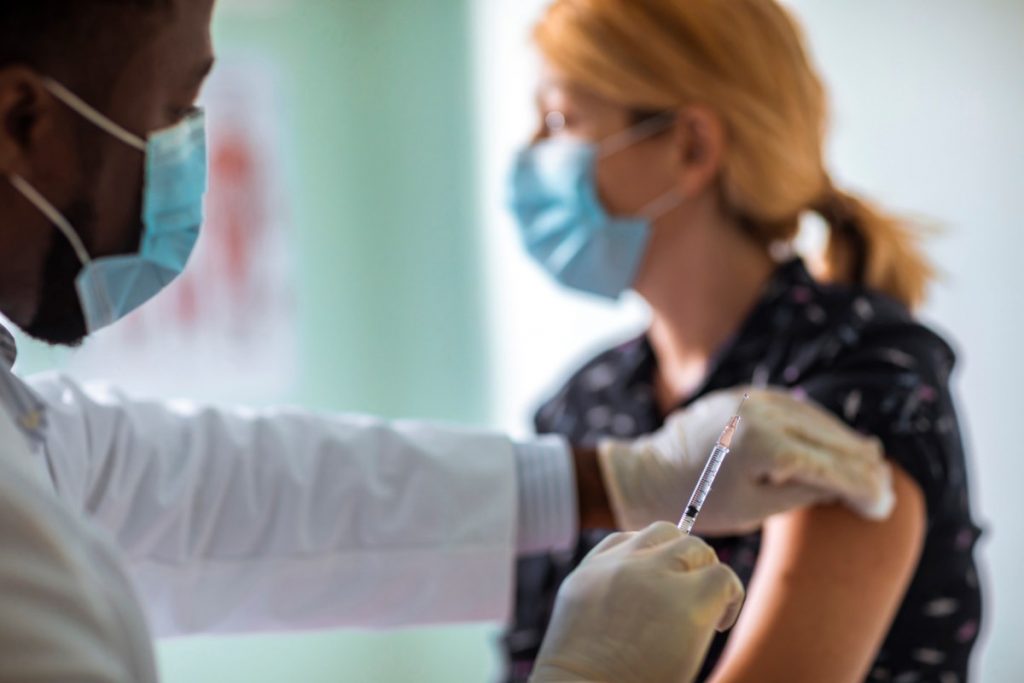
THUNDER BAY – Health authorities are preparing to ramp up the provision of COVID-19 booster shots, with all Ontario adults expected to be made eligible in the coming months.
Health Canada authorized the use of the Moderna vaccine as a booster shot for those 18 and older on Friday, days after granting similar approval to the Pfizer vaccine.
Third doses had already begun rolling out to those at higher risk of COVID-19, like people over 70, health care workers, those who are immunocompromised, and Indigenous people.
Thunder Bay’s medical officer of health, Dr. Janet DeMille, urged people in those groups to get their booster shots, noting people 80 and older are at particularly high risk.
“I certainly would encourage people to get a booster dose if they’re eligible,” she said. “One group I’m particularly concerned about is those who are older, because there is some evidence that there’s a waning of immunity [after two doses]. When there are breakthrough cases, they’re still the ones who are more likely to get a serious illness that requires hospitalization.”
Ontario’s chief medical officer of health has said there’s no firm evidence yet that adults in younger categories will face waning immunity in the same way as those 70 and up, while the National Advisory Committee on Immunization (NACI) has said booster shots aren’t needed for those who aren’t in one of the higher-risk groups.
Still, the provincial government has announced plans to make booster shots available to all adults 18 and older “over the coming months.”
DeMille said the booster shot “adds another layer of protection” for individuals, as well as communities.
“We have a good immunization rate, and we know these vaccines work really well,” she said. “But there is the risk, especially as time increases from the second dose, that [people] could experience some waning of immunity… We know the boosters do boost immunity, and in some jurisdictions have resulted in less breakthrough cases.”
While she encourages adults to get booster shots when they’re eligible, she said the need is less urgent for those outside of the higher-risk categories .
“There’s no immunological cliff where suddenly at six months, people need to get it right away,” she said. “People may be a little bit more relaxed [about booking their third dose], and I think that’s okay. Two doses work really well, but the farther you get from your second dose, the more benefit that booster dose can provide.”
The booster shots are authorized by Health Canada to be administered at least six months after an individual has completed the primary series of vaccines.
Moderna’s COVID-19 booster is a half dose of the regular vaccine, 50 micrograms. Pfizer’s booster shot is identical for that used for first and second doses.
The province has not yet announced details of its plan to expand booster doses to all adults, but DeMille said she expects that to come shortly.
“What we’re anticipating is that the province will announce the use of booster doses rolling out in January for the rest of the population. I don’t know what that will look like, whether it will be going down in ages or specific to different groups.”
Booster shots for those already eligible are being administered at the health unit’s vaccine clinic at the CLE building, as well as some local pharmacies, with demand expected to pick up over the coming weeks.
Health care organizations like the hospital will administer boosters to their own staff, DeMille said.
Indigenous partners who rolled out first and second doses are also planning clinics, while the Sioux Lookout First Nation Health Authority is coordinating Operation Remote Immunity 3.0 to reach First Nations in the north.