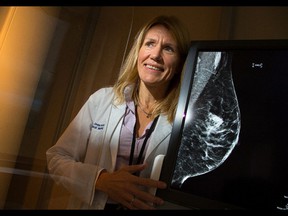
A new paper published this week in the Journal of Medical Screening suggests a Canadian study used to form breast screening guidelines around the world for decades is flawed.
The Canadian National Breast Screening Study (CNBSS) started with two breast screen trials in 1980, and eventually involved tens of thousands of women in 15 Canadian urban centres. Investigators concluded that an annual mammogram for women aged 40 to 49 who are at average risk of breast cancer does not reduce the mortality rate any more than a physical exam.
As a result, Canadian researchers concluded that annual screening in women aged 40 to 49 at average risk does not reduce breast cancer mortality any more than a physical exam. Their findings were the basis for guidelines around the world that do not recommend annual screenings for women in that age group, including the most recent set put forth by the Canadian Task Force on Preventive Health Care.
I did not expect to find such clear, clear evidence of what was going on.
The risk of false positives and over-diagnosis is cited as a key issue in earlier screening. In such cases, even benign lumps would require a biopsy. Experts argue that radiation, chemotherapy or surgical treatment of breast cancer can be harmful, and that must be considered in treating a patient who might not even have it.
Dr. Jean Seely, co-author of the new paper and a professor of radiology at the University of Ottawa and head of breast imaging at The Ottawa Hospital, told CBC News there is “conclusive confirmation” the study was conducted with women — particularly in some locations — who were not randomly assigned.
She said when the authors of the paper interviewed 28 staff members who worked on the original study, some confirmed there were multiple instances in which women with detectable lumps, who were pre-screened by a nurse, were deliberately placed in the mammography group.
Debate has swirled around the studies since the results of the CNBSS were published. Critics have questioned the inclusion of women with advanced cancers, the quality of the screening participants received, and the factors used to determine whether mammograms were beneficial for women in their 40s. Experts have also voiced doubt about the randomization, though an investigation conducted by external experts failed to find “credible evidence” that it was subverted. Those investigators further said that if there were acts of subversion, they were few and their impact on the study’s findings trivial.
Seely hopes the evidence she and her co-authors presented in their paper lead to change.
“I think probably the most personal challenging moments that I have in my practice are when I see a woman in her 40s who presents with an advanced breast cancer,” she told CBC News. Most often, the patient was told by a family doctor that she didn’t need to be screened because of her age.
“On a personal level, as a professional, I find this very, very difficult,” Seely told CBC News.
Women aged 50 and older have higher rates of breast cancer diagnosis, according to the Canadian Cancer Society, but women between 30 and 49 with breast cancer are more likely to die from it. They represent 17 per cent of all cancer deaths in Canada, compared to eight per cent of women aged 50 to 69.
The study’s lead, Dr. Anthony Miller, a professor emeritus at the Dalla Lana School of Public Health at the University of Toronto, told CBC News, “We had a great deal of understanding of what could go wrong with randomization and we took a great deal of care to ensure it didn’t apply in the National Breast Screening Study.”
Miller told CBC News extensive quality-control measures maintained the integrity of the study, and he denies the possibility that any women were not placed randomly within the trials.
Seely and her co-authors have brought their concerns about the CNBSS to the University of Toronto and the Canadian Medical Association Journal, both of which were involved in the original study.
In a statement, the university’s office in charge of research, oversight and compliance said it is reviewing their concerns, and “places a high value on research integrity.”
The university did not specify details or a timeline of its review.
In her own statement to CBC News, Dr. Kirsten Patrick, interim editor-in-chief of CMAJ, said the journal “will co-operate with the university office’s process fully and will await the office’s report on the outcome of the review before deciding if any further action is needed.”
She did not provide any further comment.