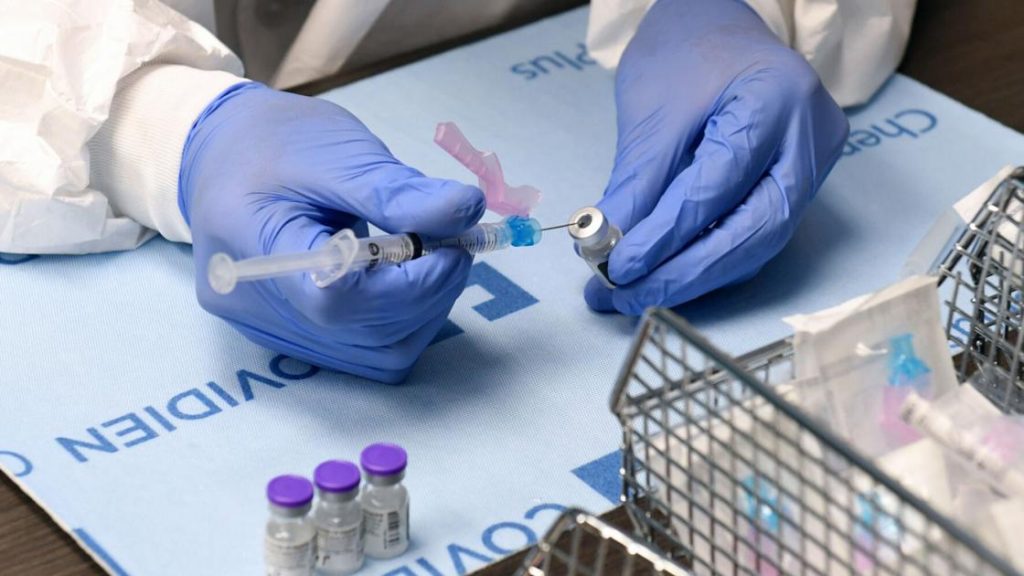
A pharmacist handles vials of the Pfizer-BioNTech COVID-19 vaccine. A New York state Supreme Court judge late Friday handed down a temporary restraining order to halt the state’s vaccine mandate that goes into effect Monday. CSEA sued the state alleging the mandate was illegally unilateral and ordered without proper bargaining. Ethan Miller/Getty Images/TNS