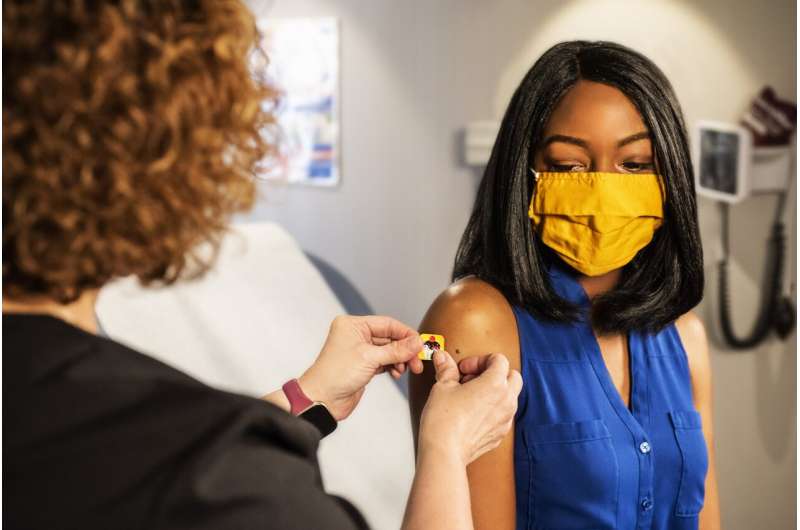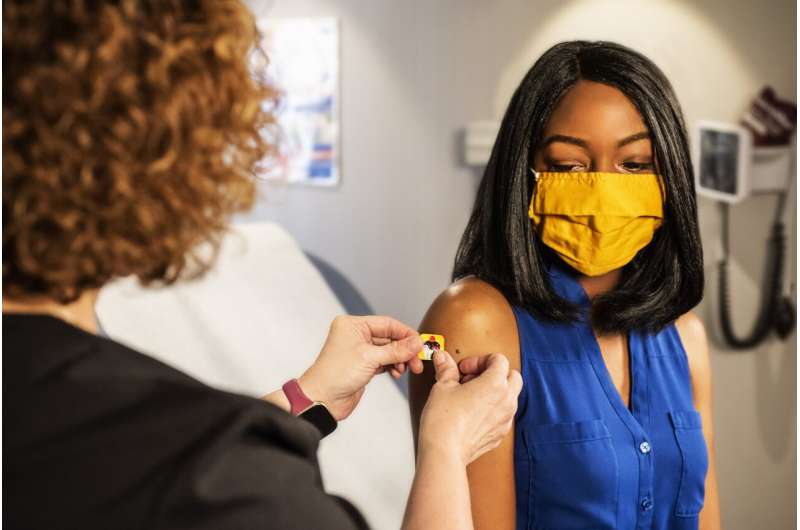
For 60 years, doctors and scientists searched for a vaccine against a common virus that, while sometimes deadly, is little known to the public. The hunt is over.
The U.S Food and Drug Administration cleared GSK Plc’s shot against respiratory syncytial virus on Wednesday. The product will go on sale in the coming weeks for older adults.
Medical breakthroughs rarely come alone, and this one is no exception. GSK, seeking to reassert its role as a key vaccine industry player after falling behind in COVID-19, will likely be forced to battle it out with one of the biggest pandemic winners, Pfizer Inc., within weeks.
“This is certainly a revolution in preventative care,” said Emily Field, head of European pharmaceutical research at Barclays Plc. GSK already sells a blockbuster vaccine to protect older adults from shingles, and if they can repeat that success in RSV, “then they’ll be in a very good position,” Field said.
Being first to market will allow GSK to get a head start on discussing its shot with doctors and insurers. AstraZeneca Plc and Sanofi are also preparing a long-acting antibody for RSV this year. And another pandemic hero, Moderna Inc., is working on a messenger RNA shot for the respiratory disease.
RSV proved “one of the more elusive vaccine targets,” Luke Miels, GSK’s chief commercial officer, said in an interview. “The world is fortunate that a number of innovations have enabled this.”
GSK called its shot Arexvy so it would sound like RSV.
Discovered in 1956, the virus quickly became recognized as one of the most common causes of childhood illness. In the 1960s, an experimental vaccine was tested in babies. But instead of protecting the infants, the shot turned out to exacerbate the disease, resulting in more hospitalizations and two deaths.
Chilling effect
The outcome had a chilling effect on the scientific community, damping research efforts for at least another decade.
RSV, characterized by an acute respiratory illness, affects an estimated 64 million people globally and causes 160,000 deaths each year, according to the U.S. National Institute of Allergy and Infectious Diseases.
The virus causes mild respiratory symptoms in healthy adults and older children. Yet for the elderly and babies, it can trigger severe infection and become life-threatening. It’s the leading cause of hospitalization for young kids in America. For severely ill patients, the only option is an approach reminiscent of the pandemic’s early days: to place them on a ventilator to help them breathe.
With four products likely reaching pharmacy shelves in the U.S. this year, drugmakers will be racing to quickly establish dominance. Two markets are emerging: people who are older or have weak immunity and pregnant women and babies.
Young and old
GSK has estimated peak sales at more then £3 billion ($3.7 billion) for its vaccine for adults 60 and above, and analysts see a potential market worth $10 billion by 2032. One of the two vaccines Pfizer plans to introduce targets the same group, as does Moderna’s experimental product.
A U.S. Centers for Disease Control and Prevention panel will meet next month to discuss both GSK and Pfizer’s shots and make recommendations, which could be key to how they’re marketed. Both vaccines have shown high efficacy in clinical trials—roughly 94% against severe disease for GSK and about 86% for Pfizer—and both can be given alongside annual influenza shots.
“This is meaningful innovation,” GSK’s Miels said. “We plan to do it justice.”
One potentially distinguishing factor could be durability. Both companies are expected to soon present data showing whether protection extended over two seasons, which normally occur during colder winter months, similar to flu.
Pfizer has another vaccine in the works that targets pregnant women, extending protection to their babies for about six months months thanks to antibodies transferred from the mother. Astra and Sanofi, meantime, will offer a passive immunization aimed at children through their first RSV season. It will be called Beyfortus, a name chosen to sound like baby fortress.
Long road
“The world’s waited for something like this for 60 years,” said Tonya Villafana, global head of infectious diseases at Astra, who has shepherded the product’s development. She hailed the impact “it could have on babies, their families and health-care systems over the next decade.”
After the 1960s research scare, scientists didn’t really advance on RSV until the late 1970s. That’s when Pfizer’s Bill Gruber, then a medical student, recalls working with a team led by a doctor named Paul Glezen on a new approach.
Glezen wondered why some babies who got RSV developed bronchiolitis and others didn’t, prompting him to study the amount of antibodies passed by their mothers via the umbilical cord.
Gruber, a student and then a resident at Baylor College of Medicine, was in charge of tracking babies showing up at the hospital with bronchiolitis so that Glezen could match them to the cord blood and its antibody content. The idea was to duplicate the levels of maternal antibodies that reduced infection.
Reaching meaningful progress took many more years, in part because of the multiple mechanisms RSV uses to evade immunity. The breakthrough came about a decade ago with a better understanding of the structure of the preF protein that the RSV virus uses to attack human cells.
But when Pfizer showed its maternal shot worked to protect babies after birth last year, Gruber, who is the company’s head of vaccines, knew there was one person he had to share the good news with. On a video call with Glezen, Gruber thanked his 92-year-old former mentor for his years of research, which helped make a sometimes fatal illness a less-threatening prospect for future generations.
2023 Bloomberg L.P.
Distributed by Tribune Content Agency, LLC.
Citation:
First RSV vaccine caps 60-year search to stem pervasive lung illness (2023, May 6)
retrieved 6 May 2023
from https://medicalxpress.com/news/2023-05-rsv-vaccine-caps-year-stem.html