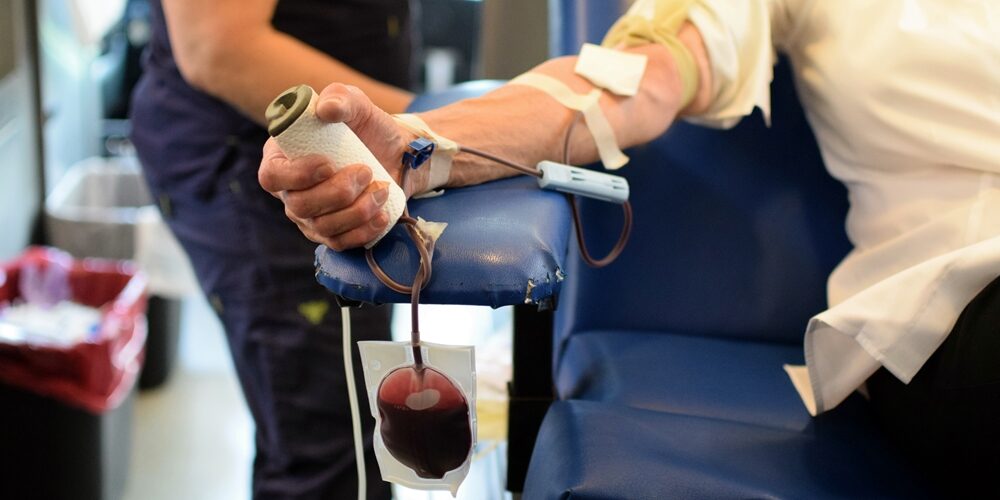
Six of 89 samples from medical equipment at long-term care facilities tested positive for the SARS-CoV-2 virus responsible for COVID-19.
The COVID-19 virus may be spreading at long-term care homes through shared medical equipment.
A Vancouver Coastal Health Authority-led study, released Friday, has found shared equipment such as blood pressure cuffs is at risk of contamination.
Vancouver Coastal Health said once it learned this information, shared blood pressure cuffs and other medical equipment at the sites were replaced with either disposable cuffs or dedicated equipment that is not shared between patients.
VCH collected samples at three long-term care homes during outbreaks of COVID-19 to better understand the extent to which environmental contamination occurs in long-term care settings.
Eighty-nine surfaces were swabbed, including entrances, kitchens, staff communal areas, nursing stations and care areas.
Six of the 89 samples tested positive for the SARS-CoV-2 virus responsible for COVID-19, VCH said in a news release Friday.
In all three facilities, the virus was detected on standard reusable blood pressure cuffs, for a total of four contaminated blood pressure cuffs of nine that were tested.
The virus was also detected on the handle of a mobile linen cart and on the touch display of an electronic tablet used for electronic medication records.
Lead author Atiba Nelson, a public health and preventive medicine resident physician who led the environmental swabbing with a VCH team, said the study was done in sites with known outbreaks and where enhanced cleaning protocols were in place.
“Although more research is needed to determine if this kind of contamination could contribute to transmission of the virus, it did highlight areas of concern,” Nelson said in a statement.
Health officials say while person-to-person transmission is believed to be the primary driver of outbreaks in long-term care facilities, the findings, published this month in the American Journal of Infection Control, suggest medical equipment is a potential route for transmission of the virus.
The authors of the paper recommend enhanced environmental cleaning for all medical equipment or prohibiting communal use of the equipment.
CLICK HERE to report a typo.
Is there more to this story? We’d like to hear from you about this or any other stories you think we should know about. Email [email protected].