Seven new cases of COVID-19 were reported in the province Friday, following news that a suspected fourth case of the coronavirus variant first reported in the U.K. has been detected in New Brunswick.
In an interview Friday morning, Premier Blaine Higgs said he doesn’t know where the variant case was located, but he was told a fourth one was found this week. This comes after the province announced Tuesday that it detected three variant cases.
“The cases that are known are isolating,” he said. “And they have had very limited contact with anyone.”
Higgs expects more cases of the variant will be detected, and he’s unsure how well it can be contained.
Chief Medical Officer of Health Dr. Jennifer Russell did not mention the suspected fourth case at Thursday’s COVID-19 news briefing. A Health Department spokesperson said the case is related to one of the first three variant cases.
“A close contact of a previous U.K. variant confirmed case has tested positive for COVID-19 and it is suspected they have the variant as well,” Bruce Macfarlane said in an emailed statement Friday morning.
He said a sample for sequencing is being sent to the National Microbiology Laboratory in Winnipeg for confirmation. In the meantime, the individual is being treated as if they have the variant.
Russell has said the arrival of the variant in New Brunswick is expected lead to larger outbreaks moving faster. It can become the dominant strain within three months. Age is the biggest risk factor.
Seven new cases reported, in three zones
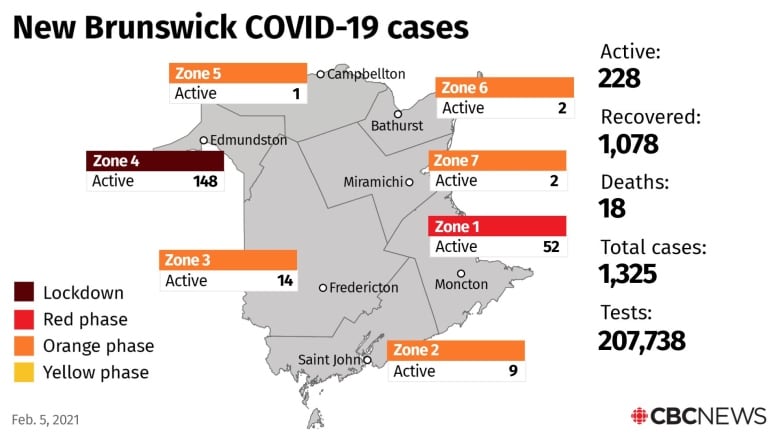
Public Health will not hold a live-streamed briefing Friday, but in a news release it said seven new cases have been recorded. The cases break down in this way:
Moncton region, Zone 1, one case:
- an individual 80-89
Fredericton region, Zone 3, one case:
- an individual 50-59
Edmundston region, Zone 4, five cases:
- an individual 20-29
- an individual 30-39
- three people 70-79
There are now 228 active cases in the province, and the number of confirmed cases so far in the pandemic is 1,325. Since Thursday, 35 people have recovered for a total of 1,078 recoveries.
There have been 18 deaths.
Six patients are in hospital, and three are in intensive care. A total of 207,738 tests have been conducted, including 1,819 since Thursday’s report.
New testing clinic opens in Campbellon
A new COVID-19 testing clinic has opened in the Campbellton Regional Hospital.
The new clinic will help with the increased demand in the region because of the mandatory weekly screening for travellers and people from border communities who are entering New Brunswick, Vitalite Health Network said in a news release Friday.
Tests are available by appointment only, by calling Tele-Care (811) or by filling out an online form on the gnb.ca website.
Vaccination of everyone could take until end of 2021
Premier Blaine Higgs says it could take until the end of 2021 for everyone in New Brunswick who wants a COVID-19 vaccine to get one.
But the premier still remains hopeful the province will see herd-immunity by September and that more vaccines will be approved before the end of the year.
“I have a degree of confidence,” he said during an interview with Information Morning Fredericton.
“Am I 100 per cent confident? No I’m not.”
Chief Medical Officer of Health Dr. Jennifer Russell says if New Brunswickers have a cold or flu symptom, it is important to get tested for COVID-19. 3:45
Higgs spoke with Prime Minister Justin Trudeau and other premiers at a meeting Thursday night where they discussed the vaccine’s availability.
Higgs said the premiers were told the country could expect to receive six million doses of the Moderna and Pfizer-BioNTech vaccines by the end of March.
But they were still left wondering about the exact timing of distribution, which Higgs says is concerning.
“We just want accuracy so we can communicate it,” he said.
Professor studies long-term effects of COVID-19
A biology professor at Mount Allison University is researching the long-term effects of COVID-19.
Vett Lloyd and her partners are asking people who have had COVID to take an online survey about their experiences.
The survey is available to everyone but is targeted at people from Ontario and New Brunswick.
Lloyd hopes the study will shed light on so called “long-haulers,” whose symptoms continue long after they contract the virus.
“It’s been coming out that some people, instead of fully recovering, remain ill to various extents . … So we’re trying to find out what their experiences are, what experiences or what it feels like to get COVID and then still be affected by it weeks, months later.”
More than 200 people have responded to the survey in its first few days.
Public exposure notification for Zone 3
Public Health has issued the following exposure notification for the Fredericton region, Zone 3:
- Carrington & Company, the gift shop at the Delta hotel at 225 Woodstock Rd., Fredericton, on Jan. 29 from 10 a.m. to 5 p.m. and on Feb. 1 from 11:30 a.m. to 5 p.m.
What to do if you have a symptom
People concerned they might have COVID-19 symptoms can take a self-assessment test online.
Public Health says symptoms shown by people with COVID-19 have included:
-
A fever above 38 C.
-
A new cough or worsening chronic cough.
-
Sore throat.
-
Runny nose.
-
Headache.
-
New onset of fatigue, muscle pain, diarrhea, loss of sense of taste or smell.
-
Difficulty breathing.
In children, symptoms have also included purple markings on the fingers and toes.
People with one of those symptoms should: